Working with flies, mice, and human cells, scientists have demonstrated that a highly conserved protein can modulate cellular senescence, potentially opening a new avenue for future therapies [1].
DNA damage and senescence
Imagine what would happen if the computer code in the software we use daily began accumulating errors with time. A somewhat similar process is going on in any living organism: despite robust repair mechanisms, DNA in our cells accumulates damage, rendering the genetic code incorrect. Sometimes, the damage is so extensive that it leads to the cell becoming senescent, which is when it stops dividing and performing many of its functions and starts emitting a mix of mostly pro-inflammatory factors: the senescence-associated secretory phenotype (SASP).
In a new study published in the journal Aging, a group of scientists from the Buck Institute for Research on Aging and the Lifespan Research Institute investigated this process in Drosophila flies, further validating their findings in mice and in human cells.
Less SASP and inflammation
Adult female flies were exposed to radiation to induce DNA damage and ultimately senescence in their guts. The researchers confirmed an increase in markers showing that DNA repair mechanisms were working overtime to cope with the damage. However, there was also a notable increase in the senescence marker β-galactosidase, showing that many cells became senescent nonetheless. A similar picture was observed in aged flies who were not subjected to radiation, as aging causes many of the same types of damage, just more slowly.
Previous research identified several Drosophila genes associated with increased gut permeability following irradiation, including meltrin. In this study, this gene’s expression was significantly lower following exposure to radiation.
To confirm the relationship, the researchers knocked down meltrin in adult flies using RNA interference and then ran a Smurf assay to assess gut permeability. This study gets its name from feeding flies blue dye, and if the intestine is damaged, the dye leaks outside, and the entire fly soon becomes blue. Knocking down meltrin in control flies caused little change, but in the irradiated group, half as many “Smurf flies” appeared. DNA repair markers and senescence markers were reduced as well.
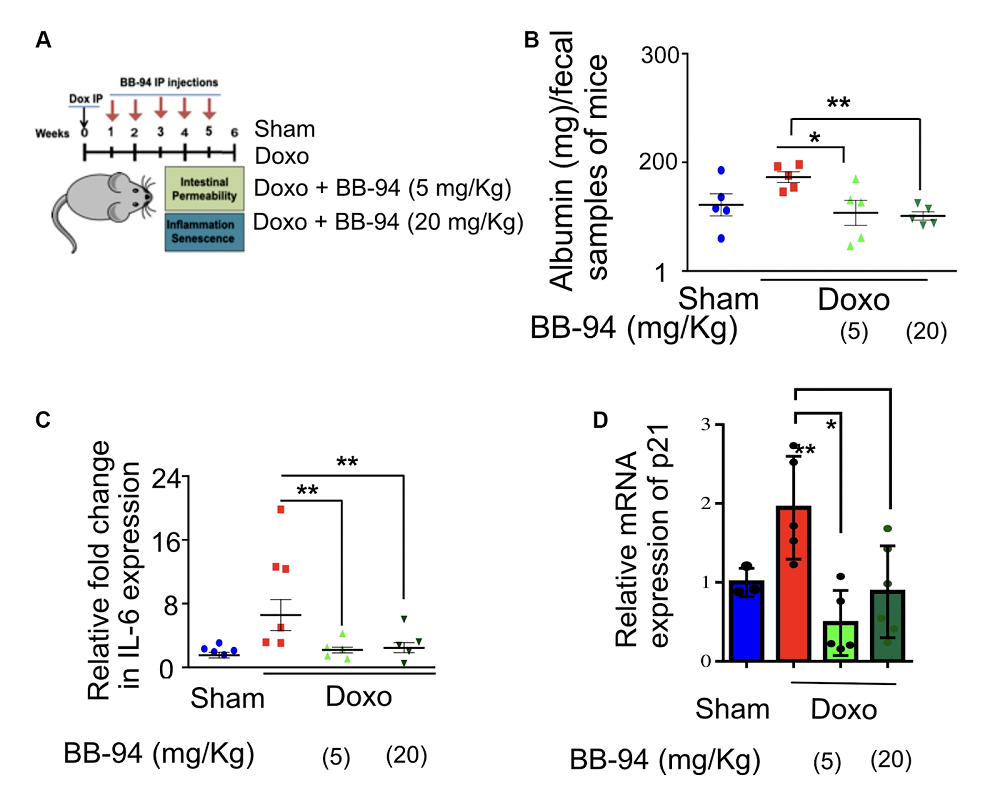
Meltrin is a highly conserved gene, and it has a mammalian homolog: ADAM19. The latter has been tied to gut function by previous research: for instance, its expression is elevated in intestinal cells in inflammatory bowel disease (IBD) [2]. To investigate further, the researchers created a mouse model by treating mice with doxorubicin, a chemotherapy drug that causes gut permeability, and blocking ADAM19 with a different molecule, batimastat (BB-94).
As expected, doxorubicin treatment increased markers of intestinal permeability (fecal albumin), inflammation (IL-6), and cellular senescence (p21) – but not in treated mice. BB-94 essentially blocked this effect, preventing increases in all three markers.
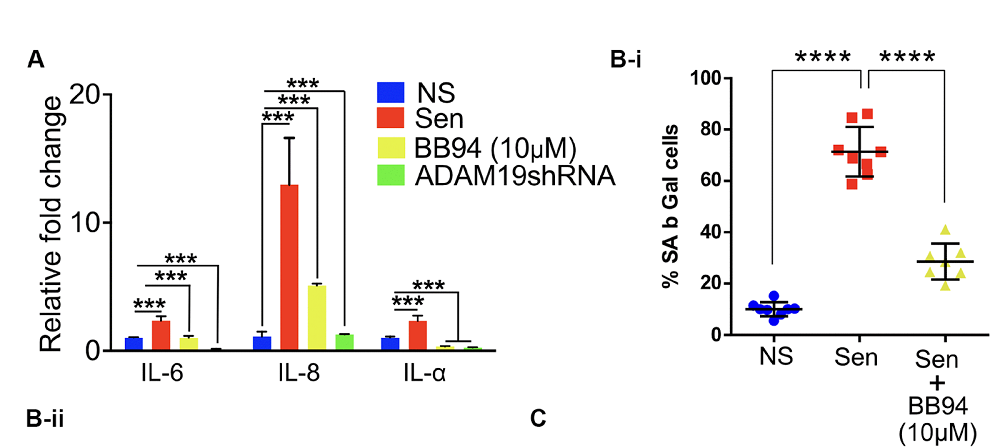
To look more closely into the effects of ADAM19 on cellular senescence, the researchers experimented with human fetal lung fibroblasts. Irradiation caused a large proportion of these cells to enter senescence and start churning out SASP-associated factors such as β-galactosidase and the pro-inflammatory cytokines IL-6, IL-8, and IL-1α. The researchers then tried either inhibiting ADAM19 with BB-94 or blocking it with an interfering RNA molecule. Both methods were effective in lowering those senescence markers.
A Disintegrin and Metalloproteases (ADAMs) are a family of enzymes that are involved primarily in ectodomain shedding: the cleavage of transmembrane proteins, which releases their extracellular domain and leaves a “stub” inside the cellular membrane [3]. Ectodomain shedding is an important element of both extracellular and intracellular signaling. Although senescent cells are suspected of tightly regulating ectodomain shedding, the exact mechanism that ties ADAM19 to a reduction in SASP factors is a subject for further studies. Still, the authors suggest that “ADAM19 inhibitors might be a novel class of senomorphics” – drugs that modify senescent cells’ behavior, making it less destructive.
Similarity between flies and mice
“It is interesting to see how a common regulatory mechanism between flies, humans, and mice is involved in senescence programming,” said Dr. Amit Sharma, head of senescence-related programs at the Lifespan Research Institute and a co-author of this study. “To me, one of the most interesting and consequential effects of cellular senescence is in the SASP expression, and now, recognizing how ADAM19, along with other ADAMs, can manipulate the secretion of SASP offers another avenue to explore senescence heterogeneity and novel therapeutic possibilities. However, these data must be tested in other senescence and aging models in the future.”
Indeed, another notable element of this study is the use of fly models with further validation in mice and human cells. The paper notes that this is fast and cost-effective. “It’s surprising to see a senescence phenotype in adult Drosophila intestine, although it has been shown in the larval developing tissue,” said Dr. Kazutaka Akagi, Assistant Professor at the Institute of Natural Medicine, University of Toyama, who was not involved in this study. “The authors’ approach that uses multiple model systems to identify a conserved function of ADAM19 proved the power of Drosophila model as a screening system for aging research.”
“It’s remarkable to see conservation of senescence-related pathways from flies to humans, the significance of which I don’t think we understand well yet,” said Dr. Pankaj Kapahi of the Buck Institute, a co-author of the study. “This discovery of senescence in flies will open new areas of investigation.”
Literature
[1] Bar, S., Hilsabeck, T. A., Pattavina, B., López-Domínguez, J. A., Basisty, N., Bons, J., Watson, M., Schilling, B., Campisi, J., Kapahi, P., & Sharma, A. (2025). Inhibition of the metalloprotease ADAM19 as a novel senomorphic strategy to ameliorate gut permeability and senescence markers by modulating senescence-associated secretory phenotype (SASP). Aging
[2] Franzè, E., Caruso, R., Stolfi, C., Sarra, M., Cupi, M. L., Ascolani, M., … & Monteleone, G. (2013). High expression of the “A Disintegrin And Metalloprotease” 19 (ADAM19), a sheddase for TNF-α in the mucosa of patients with inflammatory bowel diseases. Inflammatory bowel diseases, 19(3), 501-511.
[3] Lichtenthaler, S. F., Lemberg, M. K., & Fluhrer, R. (2018). Proteolytic ectodomain shedding of membrane proteins in mammals—hardware, concepts, and recent developments. The EMBO journal, 37(15), e99456.
View the article at lifespan.io








































