Publishing in Stem Cell Research & Therapy, researchers have found that small extracellular vesicles (sEVs) derived from young mice alleviate heart dysfunction in older mice.
Reaffirming an approach
These researchers note that there are a few ways to classify extracellular vesicles. Many of the earlier classifications were based on their origin, with microvesicles coming from the membrane and exosomes being made from encapsulated endosomes [1]. While informative, these classifications are seldom useful in practice, as current methods of separating EVs are simply based on their size. The border between small and large EVs is the 200-nanometer mark; small ones can be as low as 30 nanometers, while large ones go up to a micrometer in length.
This is far from the first laboratory success for extracellular vesicles, particularly sEVs. We have previously reported on research demonstrating that taking sEVs from young mice extends life in older mice. These researchers, however, chose to focus on one particular organ that is very often directly related to lifespan: the heart.
Partially reversing an enlarged heart
These researchers derived sEVs from adipose-derived stem cells (ADSCs): mesenchymal stem cells (MSCs) taken from the fat of mice aged 3 to 6 months. The recipient mice were almost exactly 22 months old. The mice received two doses of sEVs, with a week between the doses.
Labeling found that, after injection, sEVs migrate throughout the bodies of mice, with a notable concentration in liver tissue. However, the heart received plenty of sEVs, and they were found to migrate into the muscle tissue that provides the basic functionality of the heart.
The researchers used an ultrasound device to conduct transthoracic echocardiography, an examination process that is commonly used to diagnose heart conditions in people. Heart rate didn’t change as these mice aged, and the treatment had no effect on heart rate either. In line with previous research [2], the ability of the left ventricle to contract (systolic function) was unaffected by aging or sEVs, and in mice, the valves of the aorta tend to become calcified, resulting in higher peak velocities of blood flow [3]; unfortunately, this treatment had no effect on this aging process.
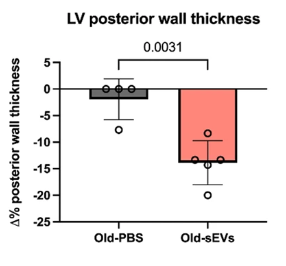
However, the treatment had a substantial effect on diastolic function: the ability of the heart to decompress between beats, which is accompanied by enlargement of the left ventricle, in both mice and people [4]. Compared to a control group, old mice treated with sEVs had significantly reduced wall thickness. The echocardiogram showed a related restoration towards youthful function in the heart, with the left ventricle becoming better at properly expanding to receive more blood. While the mice were not fully restored in this respect, the effect was statistically significant.
Tissue and metabolic changes
Aging involves significant changes to the tissue of the heart. As mice age, they grow significantly larger overall, so the researchers found no statistically significant difference between total weight and heart weight between young and old mice. The old mice treated with sEVs, however, had hearts that were significantly smaller.
A closer look found less intense fibrosis in the treated mice compared to the control group, although young mice had practically no fibrosis at all. Angiogenesis, the ability to form new blood vessels, was almost completely absent in untreated old mice but somewhat restored in the treatment group.
Other markers were even more promising. Markers of oxidative damage to both fats and proteins increase with age, but with sEV treatment, these markers were significantly reduced. Inflammatory factors related to the senescence-associated secretory phenotype (SASP) were notably decreased, as were markers of cellular senescence itself. Tissue inflammation, as measured by T cell infiltration and the DNA damage marker γH2AX were also beneficially affected by sEV treatment.
There were also positive changes to metabolic markers. Older mice accumulate metabolites that younger mice do not, such as acetyl-CoA; this accumulatedhas been associated with a failure of cellular maintenance [5]. Treatment with sEVs partially reversed this trend as well, putting the metabolome of the treated mice closer to that of the young mice.
While this study did not fully reverse aging of the heart and this is only a mouse study, it builds upon a useful body of research suggesting that sEVs may be useful in lengthening lifespan and treating age-related conditions. Cardiovascular failure is the world’s leading cause of death; if such a treatment can be brought to market, it may save many, many lives.
Literature
[1] Raposo, G., & Stoorvogel, W. (2013). Extracellular vesicles: exosomes, microvesicles, and friends. Journal of Cell Biology, 200(4), 373-383.
[2] Zhang, T. Y., Zhao, B. J., Wang, T., & Wang, J. (2021). Effect of aging and sex on cardiovascular structure and function in wildtype mice assessed with echocardiography. Scientific Reports, 11(1), 22800.
[3] De Moudt, S., Hendrickx, J. O., Neutel, C., De Munck, D., Leloup, A., De Meyer, G. R., … & Fransen, P. (2022). Progressive aortic stiffness in aging C57Bl/6 mice displays altered contractile behaviour and extracellular matrix changes. Communications biology, 5(1), 605.
[4] Paulus, W. J., & Tschöpe, C. (2013). A novel paradigm for heart failure with preserved ejection fraction: comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. Journal of the American college of cardiology, 62(4), 263-271.
[5] Eisenberg, T., Schroeder, S., Andryushkova, A., Pendl, T., Küttner, V., Bhukel, A., … & Madeo, F. (2014). Nucleocytosolic depletion of the energy metabolite acetyl-coenzyme a stimulates autophagy and prolongs lifespan. Cell metabolism, 19(3), 431-444.
View the article at lifespan.io








































